Irene Bellini (*), Paolo Bonanni (*), szczegóły Paolo Lubrano (**)(*) Department of Health Sciences, Section of Hygiene, Preventive Medicine and Health – University of Florence(**) Family pediatrician, FIMP Vaccines Networkpublished on 04/24/2017updated 08/16/2021
The flu vaccine prevents the flu and its many complications, especially in patients with compromised immune defences.
Flu vaccine
- The vaccines currently in use contain three types of viruses, and are therefore defined as trivalent.
- The viruses to be included in the vaccine are chosen from those isolated in the previous season, based on the analyzes carried out on the patients of a group of doctors, called “sentinel doctors”.
- These doctors have laboratory tests carried out on all their clients with flu symptoms, in order to isolate the circulating virus to be included in the following season’s flu vaccine.
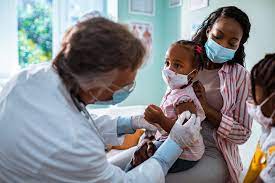 The vaccine does not prevent illnesses caused by other viruses or by flu viruses not contained in the vaccine itself. The effectiveness, i.e. the proportion of people who are protected from the flu following vaccination, is not 100 percent because there are different circulating flu viruses than those contained in the vaccine.
The vaccine does not prevent illnesses caused by other viruses or by flu viruses not contained in the vaccine itself. The effectiveness, i.e. the proportion of people who are protected from the flu following vaccination, is not 100 percent because there are different circulating flu viruses than those contained in the vaccine.
For the 2011-2012 season, the overall effectiveness measured was between 58 and 75 per cent123. Increasing the number of people vaccinated, even with a lower number, decreases the circulation of the virus.
This effect is called immunity
 The vaccine is given by intramuscular or intradermal injection. In Italy, the following types of vaccine can be used4:
The vaccine is given by intramuscular or intradermal injection. In Italy, the following types of vaccine can be used4:
Split vaccine, contains only those particles of the virus, called antigens, which stimulate the immune response and memory cells. Usually these are specific subunits that make up the outer envelope of the flu virus: hemagglutinin and neuraminidase; other fragments of internal proteins and of the capsule are also found.
- Deriving from fragmented virions, therefore with a lower content of viral components, it is less reactogenic, therefore immunogenicity and efficacy are reduced.
- Virosomal vaccine, consists of liposomes, vesicles with a lipid bilayer, which incorporate viral antigens mimicking both viral morphology and antigenic presentation.
To improve tolerability, egg proteins, nucleoproteins and matrix proteins were eliminated. The efficacy of the virosomal influenza vaccine has proved to be high in all age groups and tolerability has also allowed its use in children under 12 years of age (liposomes are the only adjuvants whose use is permitted in pediatric age).
This type of flu vaccine is able to stimulate one even in the elderly
In terms of tolerability, virosomal influenza vaccines are associated with negligible adverse reactions, lower than both the one associated with the MF59-adjuvanted vaccine and that of the whole virus influenza vaccine.
 Adjuvanted vaccine, consists of the combination of a subunit influenza vaccine with MF59, an emulsion of oil (squalene) in water containing two non-ionic surfactants. Squalene is naturally present in our body with a stimulating role in the body.
Adjuvanted vaccine, consists of the combination of a subunit influenza vaccine with MF59, an emulsion of oil (squalene) in water containing two non-ionic surfactants. Squalene is naturally present in our body with a stimulating role in the body.- This vaccine has a greater , as demonstrated by over 20 studies involving over 12,000 subjects. The benefits were more consistent in those subjects with a low response to traditional flu vaccines (elderly, transplant recipients, HIV-infected).
A study has also documented the ability of the flu vaccine to stimulate a partial defense even against strains of flu viruses significantly different from those contained in the vaccine itself. The tolerability of the influenza vaccine adjuvanted with MF59C.1 is good, even if a moderate incidence of transient local reactions is reported.
Intradermal vaccine, contains a smaller amount of antigens and, following intradermal injection, stimulates different variants than those recruited with the intramuscular vaccine. Contains no chemical adjuvants. In individuals who do not respond to the intramuscular vaccine may exhibit greater than other non-adjuvanted vaccines.
When to vaccinate
The vaccine should be administered between mid-October and the end of December. We are protected from the flu two weeks after the administration of the vaccine. This protection lasts at least a year, but even longer if you receive the adjuvanted vaccine.
How to vaccinate6
Because most of the population has most likely been infected with influenza viruses A/H3N2, A/1-11N1, and B over the past few years, a single dose of influenza vaccine is sufficient for people of all ages.
Only for children under 9 years of age who have never been vaccinated before, two doses of the vaccine are recommended, given at least four weeks apart to ensure satisfactory coverage.
The influenza vaccine is administered intramuscularly or, in the case of the intradermal vaccine, intradermally. For the intramuscular vaccine, inoculation in the deltoid is recommended for all subjects over 9 years of age; in children and infants, the recommended site is the anterolateral aspect of the thigh.
